Redox chemistry of chlorine & chloramine disinfectants
Chlorination is a very effective treatment to kill microorganisms in drinking water. Chlorine reacts with water to produce hypochlrous acid, which then dissociates to hydrogen ions and chlorate ions (see Thermochemistry of Chlorine and Chloramine). Writing the reaction as a reduction, that is electrons are added:
2HOCl +2H+ + 2e- = Cl2(aq) + 2H2O E0 = 1.609
HClO → H+ + ClO- Log K = 7.64
Depending on the pH, a system will be dominated by HOCl or by ClO-. Most distribution systems run higher than 7.5 for pH so ClO- will dominate. The disinfection process occurs through reduction of HOCl or ClO- to Cl- with coupled oxidation of organic matter
HClO + 2e- + H+ = Cl- + H2O E0 = 1.521 V
ClO- + 2e- + 2H+ = Cl- + H2O E0 = 1.747 V
From these relationships, an Eh-pH or Pourbaix diagram for the system chlorine-water can be constructed (See pdf Oxidation-reduction for technique):

Note that all chlorine reactions occur well above the stability limit for water. This is one reason chlorine is so effective as a disinfectant. It also means that chlorine in water will rather rapidly revert to chloride. Hence the importance for utilities to maintain a good chlorine residual out in the distribution system.
A downside of chlorines strong reaction with organic matter is the production of cancer-causing disinfection byproducts. Of primary concern are trihalomethanes such as chloroform. To reduce the severity of byproduct formation, many utilities substitute chloramines for chlorine.
Monochloramine is a less effective bacteriacide than chlorine, but produces far fewer toxic byproducts. It is made from the reaction of chlorine and ammonia:
NH3 (aq) + HOCl → NH2Cl + H2O
Chloramine species vary with pH, but monochloramine dominates at the higher pHs used in water distribution systems
.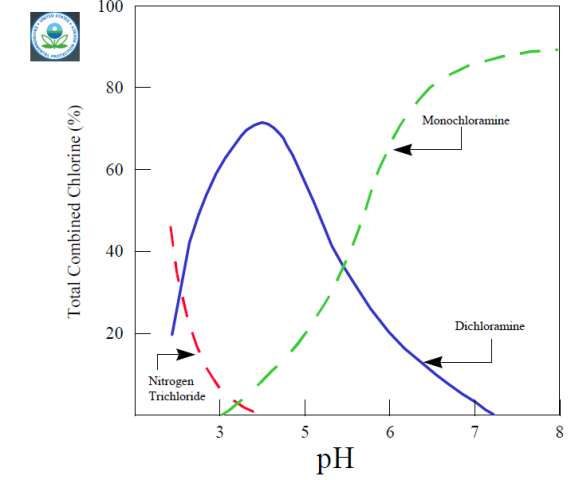
source: http://www.epa.gov/ogwdw/mdbp/pdf/alter/chapt_6.pdf
Chloramine has a lower oxidation potential than does chlorine, hence a switch to chloramine can lead to the dissolution of earlier-formed scales. For example the lead dioxide plattnerite is highly insoluble in water with free chlorine, but has appreciable solubility in water with chloramine (see Edwards and Dudi 2004). For a review of the oxidation state of various disinfectants see Copeland and Lytle 2014.

Just as with chlorine, it is possible to construct an Eh-pH diagram for the chloramines

The reactions still lie outside the stability of waterr, but at a somewhat lower Eh than chlorine. Although the stability field for monochloramine appears small compared to dichloramine, it will dominate in water distribution systems for pH values above about 5.5. The diagram also highlights the prominence of ammonia. Any reversion of the monochloramine to ammonium ion or free ammonia is likely to provoke nitrification in the distribution system, which has negative consequences for corrosion and for water quality.
Trogolo, D. and Arey, JS (2017) Equilibria and Speciation of Chloramines, Bromamines, and Bromochloramines in Water. Environmental Science & Technology 2017 51 (1), 128-140. DOI: 10.1021/acs.est.6b03219
Copeland, A., & Lytle, D. A. (2014) Measuring the oxidation-reduction potential of important oxidants in drinking water. Journal: American Water Works Association, 106(1), E10-E20.
Corrosion Scales Home |
Site Map |
|---|